 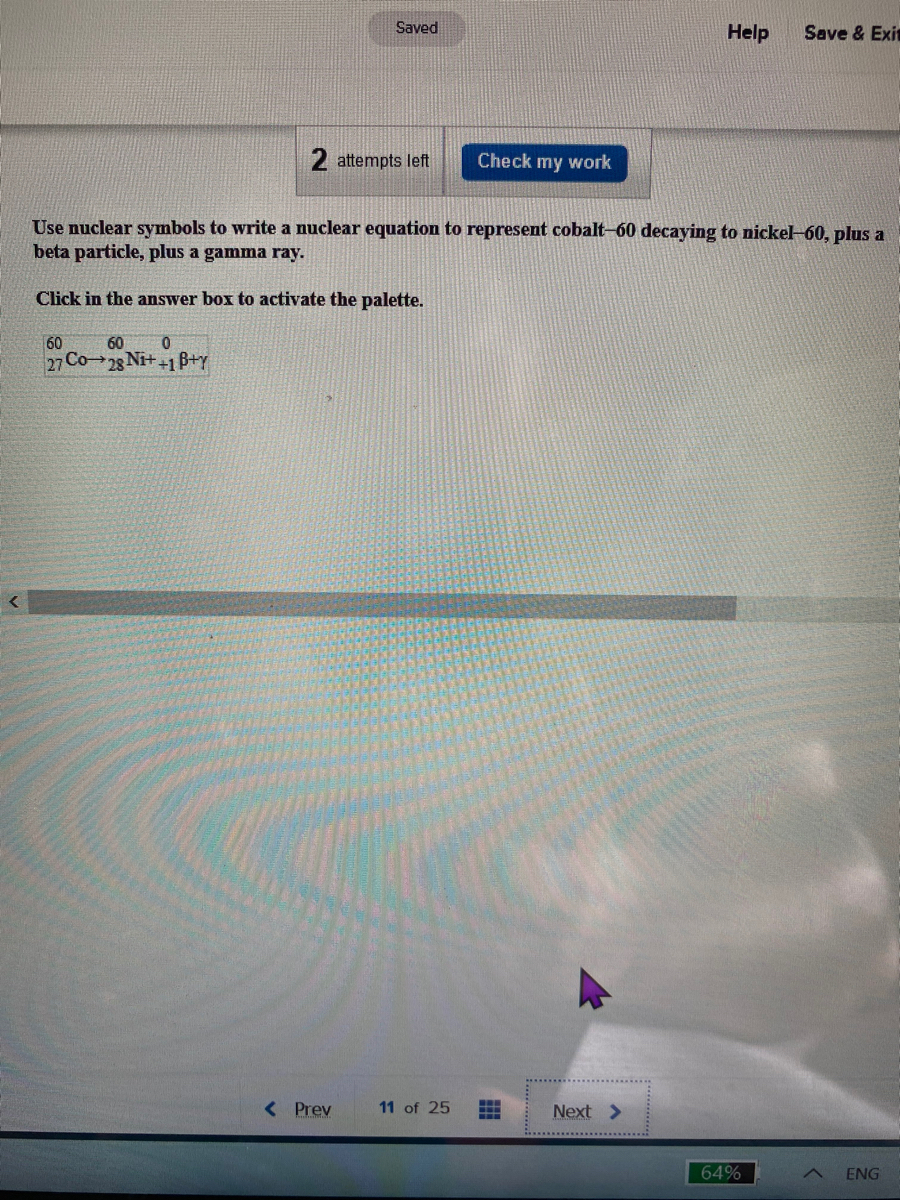
Different levels of gamma radiation produce different amounts of brightness and colors in the image, which can then be interpreted by a radiologist to reveal what is going on. This energy is detected by the scanner and converted into a detailed, three-dimensional, color image that shows how that part of the patient’s body functions. The 18F emits positrons that interact with nearby electrons, producing a burst of gamma radiation. How FDG is used by the body provides critical diagnostic information for example, since cancers use glucose differently than normal tissues, FDG can reveal cancers. (credit a: modification of work by Jens Maus)įor example, F-18 is produced by proton bombardment of 18O (818O+11p⟶918F+01n)(818O+11p⟶918F+01n) and incorporated into a glucose analog called fludeoxyglucose (FDG). The scans it produces can be used to image a healthy brain (b) or can be used for diagnosing medical conditions such as Alzheimer’s disease (c). summarizes these types of decay, along with their equations and changes in atomic and mass numbers.Ī PET scanner (a) uses radiation to provide an image of how part of a patient’s body functions. The choice is primarily due to kinetic factors, with the one requiring the smaller activation energy being the one more likely to occur. Whether electron capture or positron emission occurs is difficult to predict. This increases the n:p ratio, and the daughter nuclide lies closer to the band of stability than did the parent nuclide. Electron capture has the same effect on the nucleus as does positron emission: The atomic number is decreased by one and the mass number does not change. Like positron emission, electron capture occurs for “proton-rich” nuclei that lie below the band of stability. In most cases, the energy emitted will be in the form of an X-ray. As the outer electron drops into the vacancy, it will emit energy. The loss of an inner shell electron leaves a vacancy that will be filled by one of the outer electrons. Cobalt-60 emits γ radiation and is used in many applications including cancer treatment:Įlectron capture occurs when an inner shell electron combines with a proton and is converted into a neutron. The presence of a nucleus in an excited state is often indicated by an asterisk (*). Gamma emission (γ emission) is observed when a nuclide is formed in an excited state and then decays to its ground state with the emission of a γ ray, a quantum of high-energy electromagnetic radiation. Consequently, the n:p ratio is decreased, and the daughter nuclide lies closer to the band of stability than did the parent nuclide. Emission of an electron does not change the mass number of the nuclide but does increase the number of its protons and decrease the number of its neutrons. Such nuclei lie above the band of stability. 
The beta particle (electron) emitted is from the atomic nucleus and is not one of the electrons surrounding the nucleus. The radiation produced during radioactive decay is such that the daughter nuclide lies closer to the band of stability than the parent nuclide, so the location of a nuclide relative to the band of stability can serve as a guide to the kind of decay it will undergo ( ).ĥ3131I⟶-10e+54131Xor53131I⟶-10β+54131Xe53131I⟶-10e+54131Xor53131I⟶-10β+54131Xeīeta decay, which can be thought of as the conversion of a neutron into a proton and a β particle, is observed in nuclides with a large n:p ratio. The daughter nuclide may be stable, or it may decay itself. The unstable nuclide is called the parent nuclide the nuclide that results from the decay is known as the daughter nuclide. The spontaneous change of an unstable nuclide into another is radioactive decay. During the beginning of the twentieth century, many radioactive substances were discovered, the properties of radiation were investigated and quantified, and a solid understanding of radiation and nuclear decay was developed. Among them were Marie Curie (the first woman to win a Nobel Prize, and the only person to win two Nobel Prizes in different sciences-chemistry and physics), who was the first to coin the term “radioactivity,” and Ernest Rutherford (of gold foil experiment fame), who investigated and named three of the most common types of radiation. Describe common radiometric dating techniquesįollowing the somewhat serendipitous discovery of radioactivity by Becquerel, many prominent scientists began to investigate this new, intriguing phenomenon.Calculate kinetic parameters for decay processes, including half-life.Write and balance nuclear decay equations.Identify common particles and energies involved in nuclear decay reactions.Recognize common modes of radioactive decay.By the end of this section, you will be able to: 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
